Production has been temporarily halted while the company upgrades its sterilisation and hygiene controls to prevent future contamination.
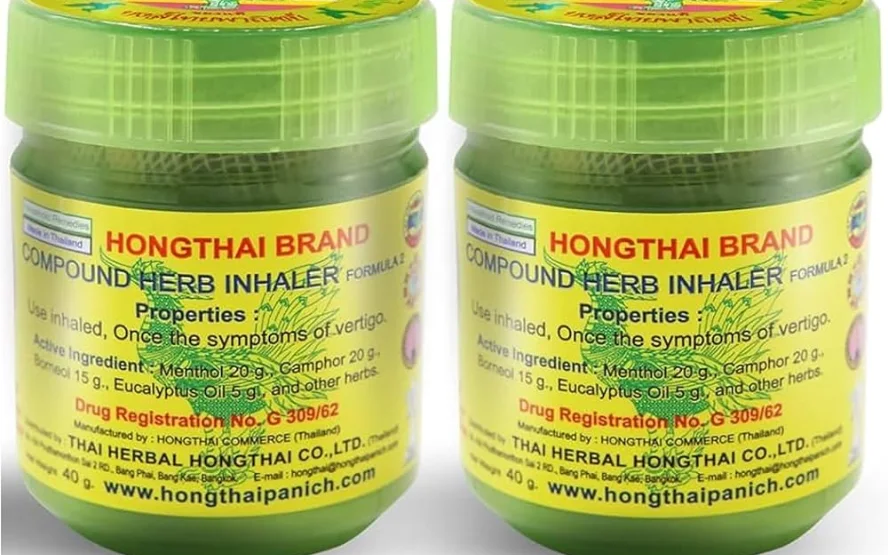
Thailand’s Food and Drug Administration (FDA) has announced a Thai herbal inhaler recall after detecting microbial contamination in a popular product.
The affected item, “Formula 2” inhaler from Hong Thai Herbal (Lot 000332, produced in December 2024), contained excessive yeast, mould, and the harmful bacterium Clostridium perfringens.
The FDA said the contamination could pose health risks, particularly for people with weakened immunity. The company confirmed that only one batch was affected and is now being withdrawn from shelves.
Consumers who purchased the product are urged to check batch numbers and stop using the affected inhalers immediately. Refunds or replacements are being offered.
Production has been temporarily halted while the company upgrades its sterilisation and hygiene controls to prevent future contamination.
At GulfRepost.com, we believe in the power of information — bringing you breaking headlines and in-depth reports that keep you connected to what’s happening across the GCC. Stay informed, stay ahead, with the most reliable Gulf news portal in the UAE.
Subscribe to Our Newsletter
Keep in touch with our news & offers
Thank you for subscribing to the newsletter.
Oops. Something went wrong. Please try again later.
G
Gulf Repost Newsletter
Your daily dose of news, trends, and updates—subscribe now!
Thank you for subscribing to the newsletter.
Oops. Something went wrong. Please try again later.
Most Viewed
1
2
3
4
5
What to read next...
September 19, 9:47 AM
September 19, 10:04 AM
September 19, 10:55 AM
September 19, 11:41 AM
September 19, 12:08 PM
September 19, 12:25 PM
September 19, 12:35 PM
September 19, 1:03 PM











